



Also in this section: Management of Rectovaginal Fistula: Colorectal Perspective

Obstetric fistulas account for most of the pelvic floor fistulas world-wide. Rectovaginal fistula (RVF) is an abnormal communication between the rectum and vagina. Injuries during childbirth are the most common cause of rectovaginal fistulas. Other causes include pelvic surgeries (including gynecological and rectal surgeries), inflammatory bowel disease, malignancy, and radiation (Kniery et al 2015).
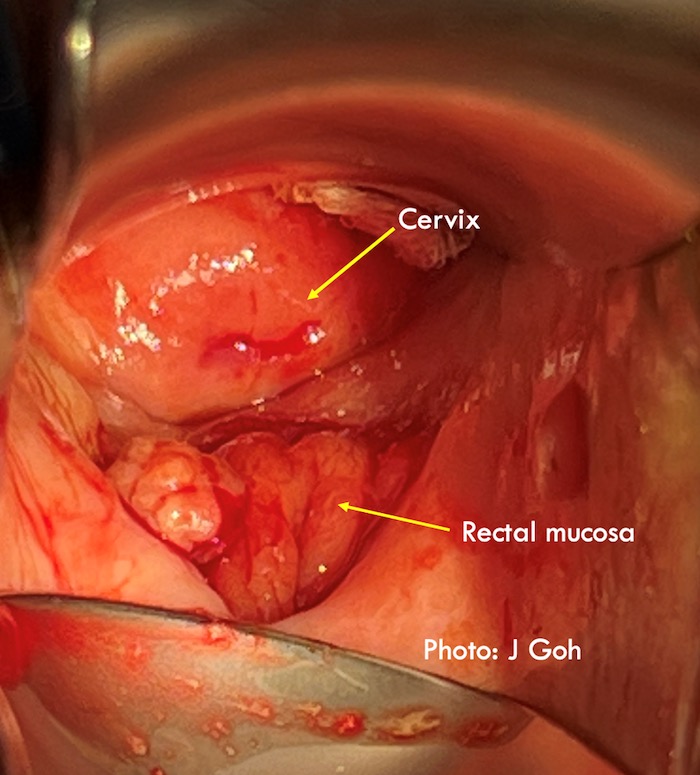
There is no standardized definition for a complex fistula. Some define a simple RVF as one that is smaller than 2.5 cm, more distal in location and resulting from trauma or infection, whereas a complex RVF is larger than 2.5 cm, results from inflammatory bowel disease/radiation/malignancy and has had failed attempts at repair (Kniery et al 2015; deBeche-Adams & Bohl 2010). However, the inclusion of location and size of the fistula in a complex fistula may also reflect the experience of the surgeon. For example, Kniery et. al (2015) would perform an abdominal repair of RVFs that are ‘near the cervix’ whereas experienced vaginal fistula surgeons may prefer the vaginal route.
Recurrent fistulas may be considered complex as they are often associated with more scarring and hence a wider dissection may be required to obtain healthier vascular tissue for fistula closure. Radiation and inflammatory fistulas are considered complex due to compromise in tissue/vascular integrity. In inflammatory bowel disease related fistulas, the advice is not to perform the fistula repair while there is active inflammation but to first optimize medical management. Compared to RVFs due to trauma, success rates for fistulas due to Crohn’s disease are significantly lower with high recurrence rates (Iglay et al 2022). Fecal diversion has not been shown to improve success rates for RVF repair (Browning & Whiteside 2015; Thayalan et al 2022; Noori 2021).
Pelvic organ mesh complications may include mesh in viscus including mesh RVF. These may be considered complex as there may be scarring due to previous pelvic surgery, the mesh may be inflamed and infected and mesh may also traverse the muscular/mucosal layers of the rectum before breaching the vagina/rectum to form the RVF. Therefore, dissection of mesh RVF may be more extensive to remove inflamed/infected mesh in the muscular/mucosal layers surrounding the RVF. Even when the mesh is not infected or in the deeper layers of the rectum, a wider dissection to remove the mesh is often required to allow a tension free closure of the fistula.
REFERENCES
Browning A, Whiteside S. Characteristics, management, and outcomes of repair of rectovaginal fistula among 1100 consecutive cases of female genital tract fistula in Ethiopia. IJGO 2015; 131: 70-73. http://dx.doi.org/10.1016/j.ijgo.2015.05.012
deBeche-Adams TH, Bohl JL. Rectovaginal Fistulas. Clin Colon Rectal Surg 2010; 23: 99-103. http://dx.doi.org/10.1055/s-0030-1254296
Iglay K, Bennett D, Kappelmamn MD, Thai S, Aldridge M, Karki C, Cook SF. A systematic review of patient burden of Crohn’s disease-related rectovaginal and anovaginal fistulas. BMC Gastroenterology 2022; 22: 36. Doi.org/10.1186/s12876-021-02079-8
Kniery KR, Johnson EK, Steele SR. Operative considerations for rectovaginal fistulas. World J Gastrointest J 2015; 7: 133-137. Doi:10.4240/wjgs.v7.i8.133
Noori IF. Rectovaginal fistulas, outcomes of various surgical repair and predictive factors of healing success, A retrospective cohort study of 40 cases. International Journal of Surgery Open May 2021;32. Doi.org/10.1016/j.ijso.2021.100335
Thayalan, K. Krause H. Goh J. A retrospective case series on transvaginal repair of rectovaginal fistula performed by a urogynaecology operative team in Australia. Aust NZ J Obstet Gynaecol 2022; 62: 263-267. Doi:10.111/ajo.13444

The gastrointestinal (GI) tract can become involved in fistula formation to several of its adjacent organs and can represent a challenge to the colorectal and gynecological surgeon. These fistulae include enterocutaneous, colovesical, and colovaginal fistulae. None are more challenging to the surgeon, however, than a perianal, anovaginal, or rectovaginal fistula. Success rates are significantly lower for these fistulas than those in the more proximal GI tract. Notwithstanding this, the anal canal and rectum are the most important parts of the GI tract and implicitly involved in function. Anal continence and bowel evacuation are achieved through the dynamic activity of this high-pressure zone in the low pelvis, with its elasticity a crucial part of each organ’s function. Both resting continence and bowel evacuation are controlled at a brainstem/subconscious level and quality of life is intimately associated with normal function; leakage, fecal urgency or defecatory difficulties are associated with significant suffering, isolation, and social stigma.
One of the difficulties in achieving high success rates in rectovaginal fistula surgery may be related to operating in a high-pressure organ, with failure driven by the dynamic forces required for the inevitable bowel motion the patient must have. As in anal fistula surgery, the goal is to cure the fistula, while at the same time preserve function; and the plethora of surgical options available speaks to the difficulties in achieving a consistent, reliable result with one operation.
"Normal" defecation and continence are generally poorly understood, with resting continence mainly contributed to by passive contraction of the puborectalis muscle, but the natural elasticity and capacitance of the rectum and the normal length of the cylindrical anal canal also contribute to prevent leakage. Understanding these mechanisms is crucial to operating in this part of the body, and being able to identify defecatory dysfunction, a common cause of excess pressure generation, may also improve healing. This is best identified preoperatively and addressed to improve the success of surgery.
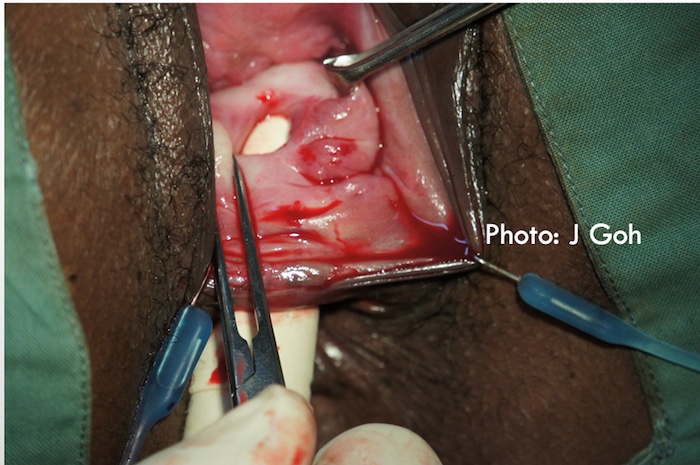
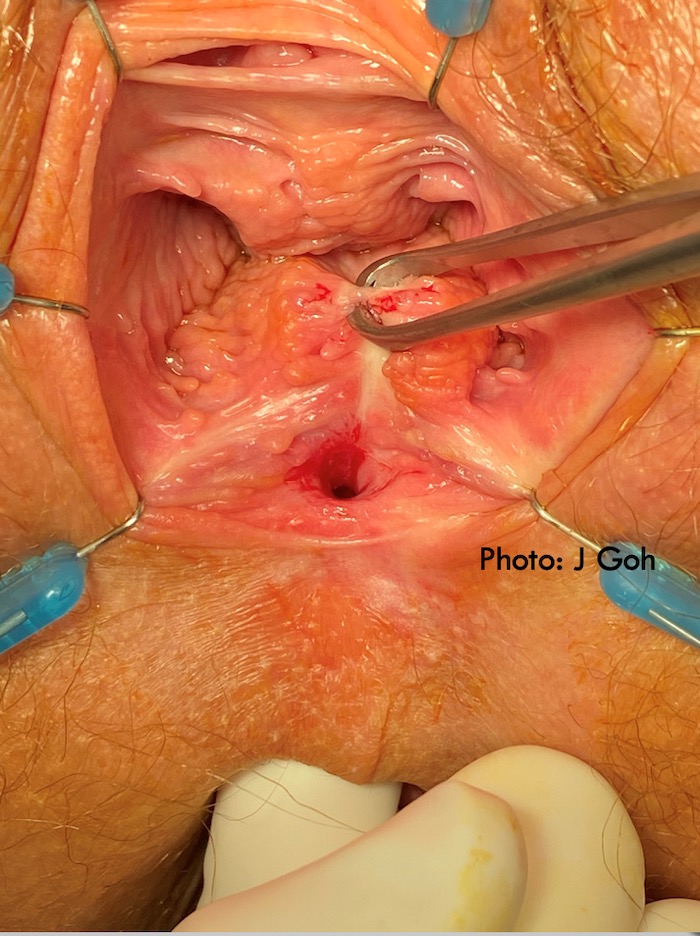
From a colorectal perspective, the principles of treatment of a rectovaginal fistula include determining preoperative continence and ensuring optimal operating conditions, optimizing tissue quality, general health, and nutritional status. Liberal use of EUA (examination under anesthesia) to assess the fistula carefully and understand its anatomy in relation to the sphincter is crucial. Failure to identify the internal opening is a clear predictor of failed surgery.
It is also important to keep the rectal pressure down, as the recurrence is likely to be derived from pressure on the rectal side of the repair. Hard or firm stool should be avoided; this can be achieved by preoperative bowel preparation and postoperative high-dose laxatives.
Overall, there is poor quality evidence in the literature regarding rectovaginal fistula repair and a previous systematic review failed due to limited availability of high-quality prospective studies, with success rates ranging from 0 to 80%.
In Crohn's disease, liaison with the treating gastroenterologist is crucial and repair should only be attempted when Crohn's disease is medically controlled and in remission. It is important to be aware of medical options including biologic therapy, with a 50% rate of healing achieved with Infliximab in the ACCENT-2 trial.